EIA: Equine Infectious Anemia
Equine Infectious Anemia (EIA) or swamp fever is a chronic infectious disease of equids caused by a lentivirus of the Retroviridae family. This virus is transmitted mechanically by hematophagous biting insects, which act as vectors of the disease. Stable flies (Stomoxys calcitrans) are capable of transmitting the infection; however, the most effective vectors are biting flies from the Tabanidae family. A vertical transmission (from the mare to the fetus in utero) as well as the transmission in the milk to nursing foals have also been described. The risk of vertical transmission is more likely if the mare presents with clinical signs before foaling. There is also a iatrogenic transmission by the use of contaminated needles or veterinary/dental equipment as well as by blood transfusions.
After the infection most horses become asymptomatic carriers of the virus, which remains latent until the horse has a stress (other diseases, severe stress, intense exercise) resulting in recurring episodes of fever, anemia, weight loss and depression. The acute form is characterised by non-specific signs such as fever, icterus, ventral edema, blood-stained feces and petechiae on the mucus membranes. In horses, EIA virus persists in blood leukocytes for life, and also occurs in plasma during febrile episodes.
Situation in Spain
Spain was free from EIA since 1983, when the last EIA outbreak was declared resolved. Since then, the only study showing EIA seroprevalence data in Spain was carried out in 2011-2013 by Cruz et al.; they analysed 555 Spanish Purebred horses in central Spain and did not detect any seropositive.
How to diagnose Equine Infectious Anemia
The diagnosis is often carried out by using two serological techniques: Enzyme-linked immunoassay (ELISA) and Agar Gel Immunodifusion Test (AGID or Coggins test).
COGGINS TEST
According to the OIE, Coggins test is the official, gold standard test for the diagnosis of EIA. It can detect antibodies 2-3 weeks after the infection, although some cases of seronegative horses 60 days post-infection have been described. Results are available after 48 hours and the interpretation of results require experience.
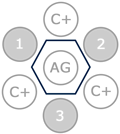
Coggins test is an immunoprecipitation where the EIA antigen is tested against the antibodies present in the horse serum and the antibodies present in a positive control serum.
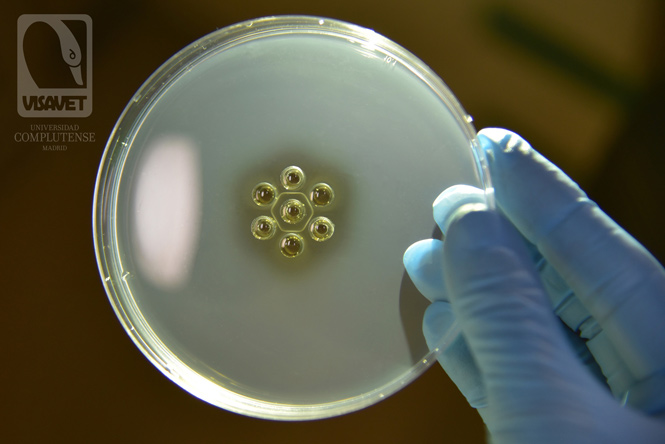
Positive AGID or Coggins test
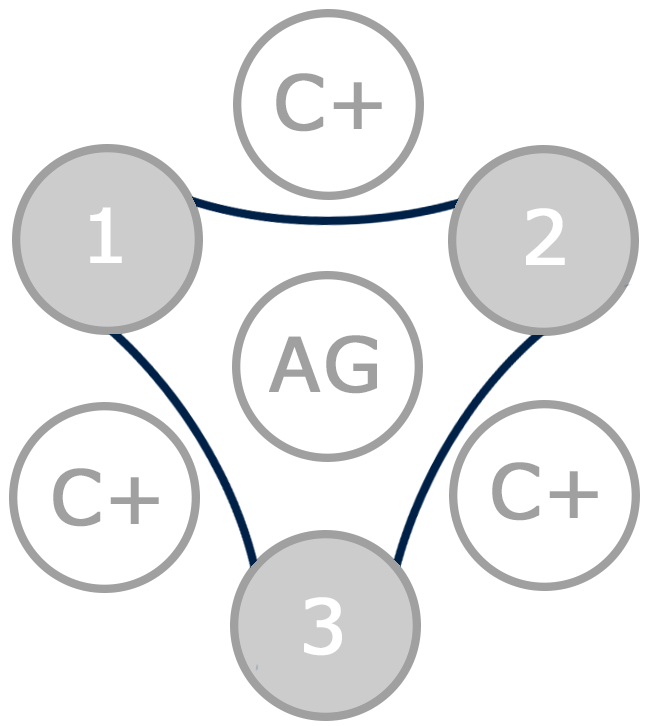
In a positive Coggins test, an immunoprecipitation occurs due to the binding of the antigen (central well) and the antibodies in the positive controls (C+) and the samples (1, 2 and 3). In the picture, the three samples (1, 2 and 3) are positive to EIA and therefore a line can be seen in all wells, giving the appearance of a hexagon.
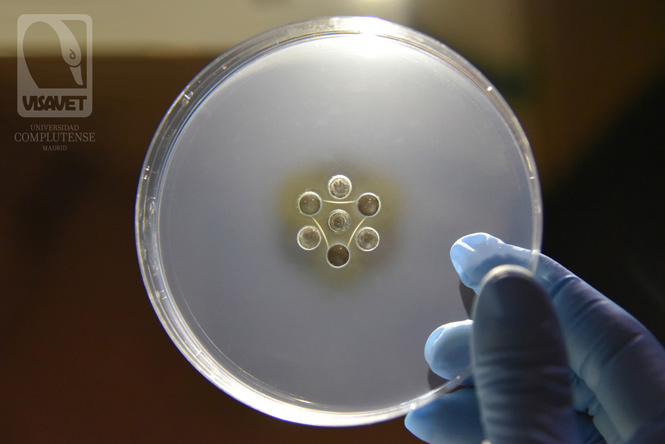
Negative AGID or Coggins test
In a negative Coggins test, an immunoprecipitation occurs due to the binding of the antigen (central well) and the antibodies in the positive controls (C+), but the immunoprecipitation does not happen in the samples (1, 2 and 3). In the picture, the three samples (1, 2 and 3) are negative to EIA and therefore a line can only be seen in the wells containing positive controls, giving the appearance of a triangle.
ELISA TEST
The ELISA test provides results in less time and with a higher sensibility and objectivity than Coggins test. Since the ELISA is more sensitive than Coggins, false positives are more likely to occur. For this reason, positive results on ELISA are confirmed with the AGID test.
RT-PCR
Reverse-transcriptase polymerase chain reaction (RTPCR) assays can also be used in the blood of suspicious horses. These tests are valuable in determining the infection status in the following cases:
- To supplement or confirm serological tests
- When there are conflicting results or when an infection is suspected but serology is negative or equivocal.
- To determine the infection status of foals born to infected mares.
Equine Infectious Anemia treatment and control
EIA is included in the list of the OIE notifiable diseases and it must also be notified to the EU health authorities. In Spain, as specified in the law 8/2003 for Animal Health, whenever there is a suspicion of EIA, the veterinary surgeon must communicate the suspicion to the official services of his/her autonommous community, who will draw the appropriate samples in order to confirm the suspicion of disease in the National Reference Laboratory. In case of a EIA seropositive found in a sample from an asymptomatic horse and tested in a private Laboratory (e.g. for a pre-exportation analysis), both the laboratory and the veterinary surgeon who sent the samples are obliged to notify the seropositive result to the authorities.
In many countries, including Spain, the culling of EIA seropositive horses is mandatory. These horses must be isolated from the rest of horses and a paliative treatment must be implemented until the culling is arranged. If an outbreak is declared, the horses in the yard must be confined in small groups, and the movement of horses on/off the premises should be stopped. Fumigation and the use of repellents against insects can aid to stop the transmission.
There is no available vaccine against EIA due to the virus mutation capacity, so the control strategies are based in setting up quarantines as well as carrying out epidemiological surveys in order to determine the existence of carriers within the horses in contact with the positive cases.
How to send us samples for informative analyses
The Equine Health Surveillance Unit (SEVISEQ) from the VISAVET centre is a Laboratory authorised for the diagnosis of EIA by ELISA and Coggins test in samples for informative analyses (prior to exportation or movements and from horses without a suspicion of EIA and without clinical signs).
In order for us to carry out these tests, serum samples must be identified with the transponder number or UELN number as well as the number of the premises where the horse resides.
For further information and the consultation of questions regarding EIA please contact us:
VISAVET Health Surveillance Centre
Complutense University Madrid (Spain)
Tel.: (+34) 913944096
seviseq@ucm.es
Bibliography
- Anon. 2009. Equine Infectious Anemia. The Center for Food Security and Public Health and Institute for International Cooperation in Animal Biologics, Iowa State University..
- Equine infectious anemia. OIE Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. 2013. Chapter 2.5.6.
- Pearson, J. E. and Knowles, R.C. 1984. Standardization of the equine infectious anemia immunodiffusion test and its application to the control of the disease in the United States. Journal of the American Veterinary Medicine Association. 184(3):298-301.
- Coggins, L., Norcross, N.L. and Nusbaum, S.R. 1972. Diagnosis of equine infectious anemia by immunodiffusion test. American Journal of Veterinary Reseach. 33(1):11-18.
- Cook, R. F., Cook, S.J., Li, F.L., Montelaro, R.C. and Issel, C.J. 2002. Development of a multiplex real-time reverse transcriptase-polymerase chain reaction for equine infectious anemia virus (EIAV). Journal of Virological Methods. 105(1):171-179.
- Cruz F., Fores P., Ireland J., Moreno MA. and Newton R. 2015. Freedom from equine infectious anaemia virus infection in Spanish Purebred horses. Veterinary Record Open. 2:e000074.2015.
- Issel, C. J. and Cook, R.F. 1993. A review of techniques for the serologic diagnosis of equine infectious anemia. Journal of Veterinary Diagnostic Investigation. Journal of Virological Methods. 5(1):137-141.
- Issel, C. J., Rushlow, K., Foil, L.D. and Montelaro, R.D. 1998. A perspective on equine infectious anemia with an emphasis on vector transmission and genetic analysis. Vet Microbiol. 17(3):251-286.
- Issel, C. J., Scicluna, M.T., Cook, S.J., Cook, R.F., Caprioli, A., Ricci, I., Rosone, F., Craigo, J.K., Montelaro, R.C. and Autorino, G.L. 2013. Challenges and proposed solutions for more accurate serological diagnosis of equine infectious anaemia. The Veterinary Record. 172(8):210.
- More, S. J., Aznar, I., Bailey, D.C., Larkin, J.F., Leadon, D.P., Lenihan, P., Flaherty, B., Fogarty, U. and Brangan, P. 2008. An outbreak of equine infectious anaemia in Ireland during 2006: investigation methodology, initial source of infection, diagnosis and clinical presentation, modes of transmission and spread in the Meath cluster. Equine Veterinary Journal. 40(7):706-708.
- Nagarajan, M.M. and Simard, C. 2001. Detection of horses infected naturally with equine infectious anemia virus by nested polymerase chain reaction. Journal of Virological Methods. 94(1-2):97-109.
- Sellon, D.C. 1993. Equine infectious anemia. Veterinary Clinics of North America Equine Practice. 9(2):321-336.
- Singha, H., Goyal, S.K., Malik, P., Khurana, S.K. and Singh, R.K. 2013. Development, evaluation, and laboratory validation of immunoassays for the diagnosis of equine infectious anemia (EIA) using recombinant protein produced from a synthetic p26 gene of EIA virus. Indian Journal of Virology. 24(3):349-356
- Soutullo, A., Verwimp, V., Riveros, M., Pauli, R. and Tonarelli, G. 2001. Design and validation of an ELISA for equine infectious anemia (EIA) diagnosis using synthetic peptides. Veterinary Microbiology. 79(2):111-121.