Most appropriate samples to be taken when facing a suspicion of infectious disease in horses
1>Whenever there is a suspicion of an infectious disease in a horse, it is important to take into account several basic aspects of the sampling for the laboratory analysis in order to avoid mistakes in this stage which could lead to false conclusions drawn from the interpretation of results:
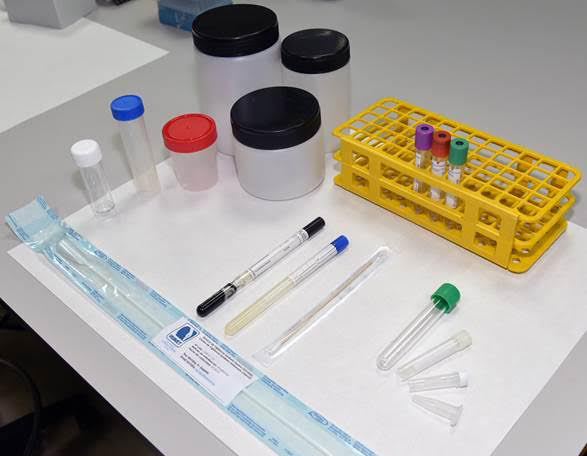
- Choosing the right material when carrying out the sampling.
- Sampling in clean/sterile conditions depending on the case.
- The samples taken for the detection of the agent or the antigen should be transported to the laboratory refrigerated.
- The samples taken for the detection of antibodies (serology) can be transported to the laboratory without refrigeration.
- In order to avoid mistakes it is very important to do a correct identification of the samples.
- In order to avoid delays in the start of the analysis, it is essential to enclose a submission form.
In this article we summarise the most appropriate samples that should be taken when facing a horse with a suspicion of infectious disease.
Equine Rhinopneumonitis (Equine Herpesvirus type 1 / type 4)

Respiratory disease
- Nasopharyngeal swab in a sterile tube with sterile saline (PCR).
- Heparinised blood: Ideally 30 ml, mínimum 10 ml (PCR).
- Serology: Complement Fixation Test, paired samples, only in non-vaccinated horses and non-vaccinated in-contact horses.
- Nasopharyngeal swab in a sterile tube with sterile saline (PCR).
- Heparinised blood: Ideally 30 ml, minimum 10 ml (PCR).
- Cerebrospinal fluid: Minimum 1 ml in a heparin tube (PCR).
- Death/Euthanasia: Brain and spinal cord (PCR, Immunohistochemistry) in a sterile container. The same in 10% formaline for histopathology.
- Serology: Complement Fixation Test, paired samples, only in non-vaccinated horses and non-vaccinated in-contact horses.
- Samples from placenta: Cervical star, body and both uterine horns in a sterile container. The same in 10% formaline for histopahology.
- Samples from fetus: Liver, lung, spleen, adrenal gland and thymus in a sterile container. The same in 10% formaline for histopahology.
- Serology: Not useful (mare, fetus). Complement Fixation Test, paired samples, only in non-vaccinated horses and non-vaccinated in-contact horses.
Neonatal disease/death
- Neonatal disease: Nasopharyngeal swab in a sterile tube with sterile saline (PCR). Heparinised blood: Ideally 30 ml, mínimum 10 ml (PCR).
- Neonatal death: Liver, lung, spleen, adrenal gland and thymus in a sterile container. The same in 10% formaline for histopahology.
- Serology: Not useful (mare, fetus). Complement Fixation Test, paired samples, only in non-vaccinated horses and non-vaccinated in-contact horses.
Equine Viral Arteritis

Respiratory disease / pyrexia / oedema
- Nasopharyngeal swab in a sterile tube with sterile saline (PCR).
- Heparinised blood: Ideally 30 ml, mínimum 10 ml (PCR). Serology: Seroneutralisation, paired samples.
Aborto
- Samples from placenta: Cervical star, body and both uterine horns in a sterile container. The same in 10% formaline for histopahology.
- Samples from fetus: Liver, lung, thymus, peritoneal fluid, pleural fluid in a sterile container. Liver, lung, thymus in 10% formaline for histopahology.
- Serology: Rarely useful (mare). Seroneutralisation, paired samples.
Neonatal death / pneumonia
- Neonatal pneumonia: Nasopharyngeal swab in a sterile tube with sterile saline (PCR). Heparinised blood: Ideally 30 ml, mínimum 10 ml (PCR).
- Neonatal death: Liver, lung, thymus, peritoneal fluid, pleural fluid in a sterile container. Liver, lung, thymus in 10% formaline for histopahology.
- Serology: Rarely useful (mare, foal). Seroneutralisation, paired samples. Better in the in-contact horses.
Detection of carrier stallions
- Serology: Screening by ELISA, confirmation of positive/equivocal by Seroneutralisation.
- Semen: Positive to Seroneutralisation. Rich fraction of the ejaculate. Refrigerated, mínimum 1 ml. PCR or Viral Isolation.
West Nile Virus
Neurological disease
- Ideally: Serum → IgM ELISA. Paired samples.
Cerebrospinal fluid → IgM ELISA, PCR. - Heparinised blood → PCR [very short viremia (2-3 days)].
- Death/euthanasia: Cerebrospinal fluid, brainstem, brain, spinal cord → PCR.
![]() If positive → It is mandatory to declare
If positive → It is mandatory to declare
Equine Influenza
Acute disease
- Nasopharyngeal swab in a sterile tube with sterile saline (PCR / NP ELISA).
- Serology: Hemagglutination inhibition. Paired samples [not useful in horses that have been recently vaccinated (< 4 months)].
Equine Infectious Anemia
Acute / Subacute / Chronic disease
- Serology: ELISA. If positive → Agar Gel Immunodifusion Test (Coggins Test).
![]() If positive → It is mandatory to declare
If positive → It is mandatory to declare
Equine Strangles (Streptococcus equi subsp. equi)
Acute disease
- Nasopharyngeal swab in a sterile tube with sterile saline (culture + PCR).
- Swab / aspirate of an abscessed lymph node (culture + PCR).
Carrier detection
- 3 nasopharyngeal swabs: Taken in 3 consecutive weeks (culture + PCR).
- Guttural pouches wash (culture + PCR).
- Serology: ELISA in order to detect affected horses in contac (subclinical disease), new horses in a premises (quarantine).
Rhodococcus equi
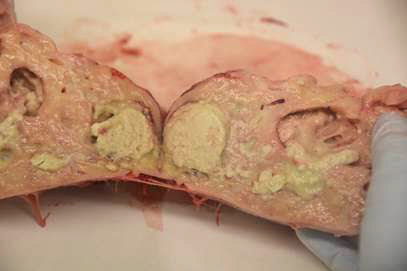
Acute disease
- Tracheal wash in a sterile tube (culture + vap A PCR).
Death / euthanasia
- Tracheal swab, lung, intestine / intestinal content (culture + vap A PCR).
Serology
- ELISA in order to detect the presence of Rhodococcus equi infection in a stud farm.
Equine Piroplasmosis (Theileria equi, Babesia caballi)
Acute disease
- EDTA blood → PCR.
- Serology: competition ELISA / IFAT in paired samples better than Complement Fixation Test.
Detection of carriers
- Serology (competition ELISA) → Positive → PCR in EDTA blood / in vitro culture.
Equine Salmonellosis (Salmonella spp.)
- Fecal samples taken from rectum and refrigerated. Ideally 3 samples → Specific culture for Salmonella.
Enterocolitis caused by Clostridium perfringens
- Fecal samples taken from rectum and refrigerated → ELISA for the detection of Clostridium perfringens enterotoxin.
A positive culture does not involve that Clostridium perfringens is the cause.
Enterocolitis caused by Clostridium difficile
- Fecal samples taken from rectum and refrigerated → ELISA for the detection of Toxins A and B of Clostridium difficile
A positive culture does not involve that Clostridium difficile is the cause.
Other cases
Finally, in those cases when there is not a clear suspicion of a particular agent, it is essential to do a correct interview in order to gather the clinical history and determine if there are any other cases in the same premises or in nearby premises. Furthermore, it is important to know the recent movements in the premises: new entries, outings to competitions, as well as any changes in the management or the feed. A list of differential diagnoses should be done; if even with all the gathered information the case is not clear and there may be several agents causing the clinical signs, the ideal samples for a diagnosis would be the following: Nasopharyngeal swab, heparinised blood and serum. If the horse has died or if it has been euthanised, the ideal samples to be collected in the necropsy (fresh tissues and tissues in formaline 10%) are: lung, liver, thymus, spleen, lymph node, and brain, cerebrospinal fluid and spinal cord if the horse presented with neurological signs.
Equine Health Surveillance Unit
VISAVET Health Surveillance Centre
Complutense University Madrid (Spain)
Tel.: (+34) 913943999
seviseq@ucm.es
Bibliography
- Cruz F. 2015. Equine Strangles. VISAVET Outreach Journal. VISAVET Health Surveillance Centre (U.C.M.).
- Burgess, B.A., Morley, P.S. 2014. Managing Salmonella in equine populations. Vet Clin North Am Equine Pract. 30(3): 623-640.
- Diab, S.S., Kinde, H., Moore, J., Shahriar, M.F., Odani, J., Anthenill, L., Songer, G., Uzal, F.A. 2012. Pathology of Clostridium perfringens type C enterotoxemia in horses. Vet Pathol. 49(2):255-263.
- Diab, S.S., Songer, G., Uzal, F.A. 2013. Clostridium difficile infection in horses: a review. Vet Microbiol. 167(1-2):42-49.
- Giguère, S., Cohen, N.D., Chaffin, M.K., Slovis, N.M., Hondalus, M.K., Hines, S.A., Prescott, J.F. 2011. Diagnosis, treatment, control, and prevention of infections caused by Rhodococcus equi in foals. J of Vet Int Med. 25 (6): 1209-1220.
- Equine infectious anemia. OIE Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2013. Chapter 2.5.6.
- Equine influenza. OIE Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2013. Chapter 2.5.7.
- Equine piroplasmosis. OIE Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2013. Chapter 2.5.8.
- Equine rhinopneumonitis. OIE Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2013. Chapter 2.5.9.
- Equine viral arteritis. OIE Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2013. Chapter 2.5.10.
- Waller, A.S. 2013. Strangles: Taking steps towards eradication. Vet Microbiol. 167(1-2): 50-60.


